target product profile fda guidance
4.9 (552) · $ 12.50 · In stock

FDA 2011 Process Validation Guidance: Lifecycle Compliance Model
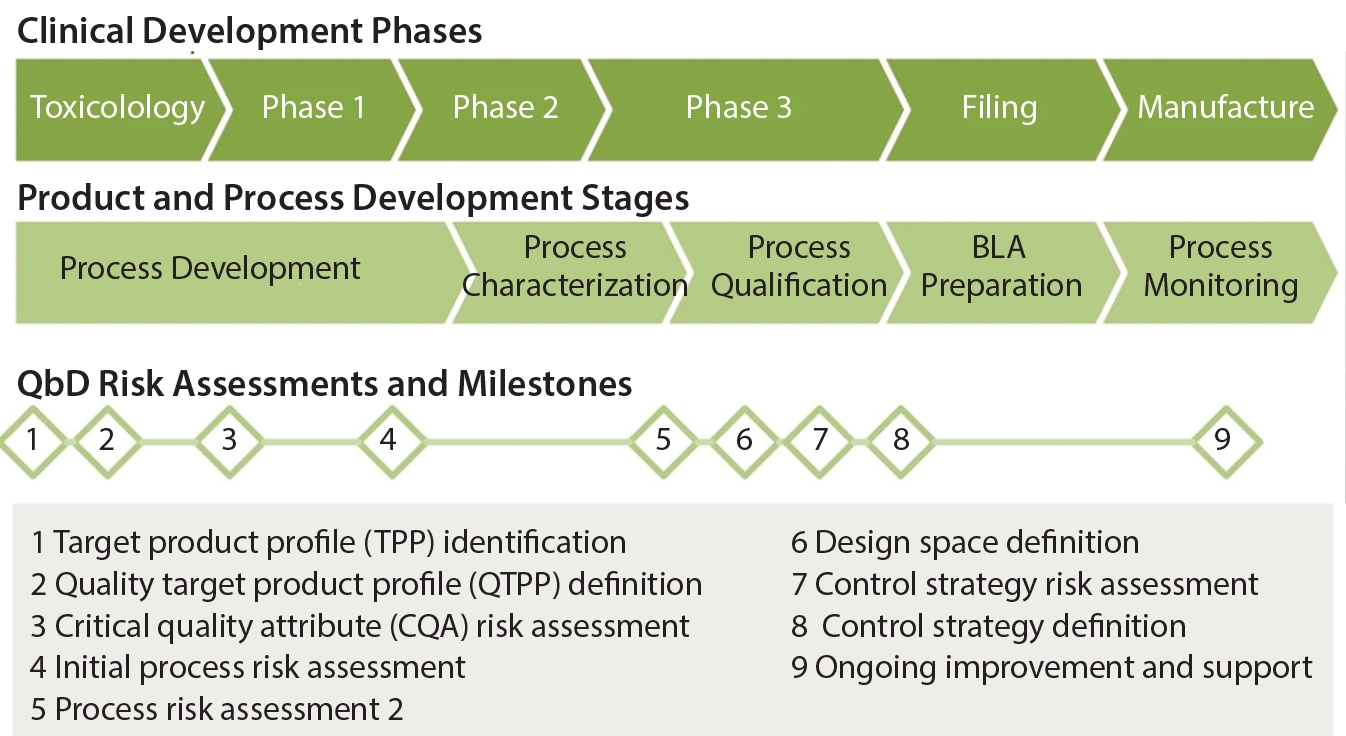
Quality By Design for Monoclonal Antibodies, Part 1: Establishing the Foundations for Process Development

Target Product Profile

The Target Product Profile

Road to commercial success –the target product profile basics part

image.slidesharecdn.com/roadtocommercialsuccessthe

The First Step for Streamlined CMC Development: Optimize the Target Product Profile < Premier Research

Injectable Combination Product Development: Facilitating Risk

FDA Guidance Target Product Profile, PDF, Food And Drug Administration
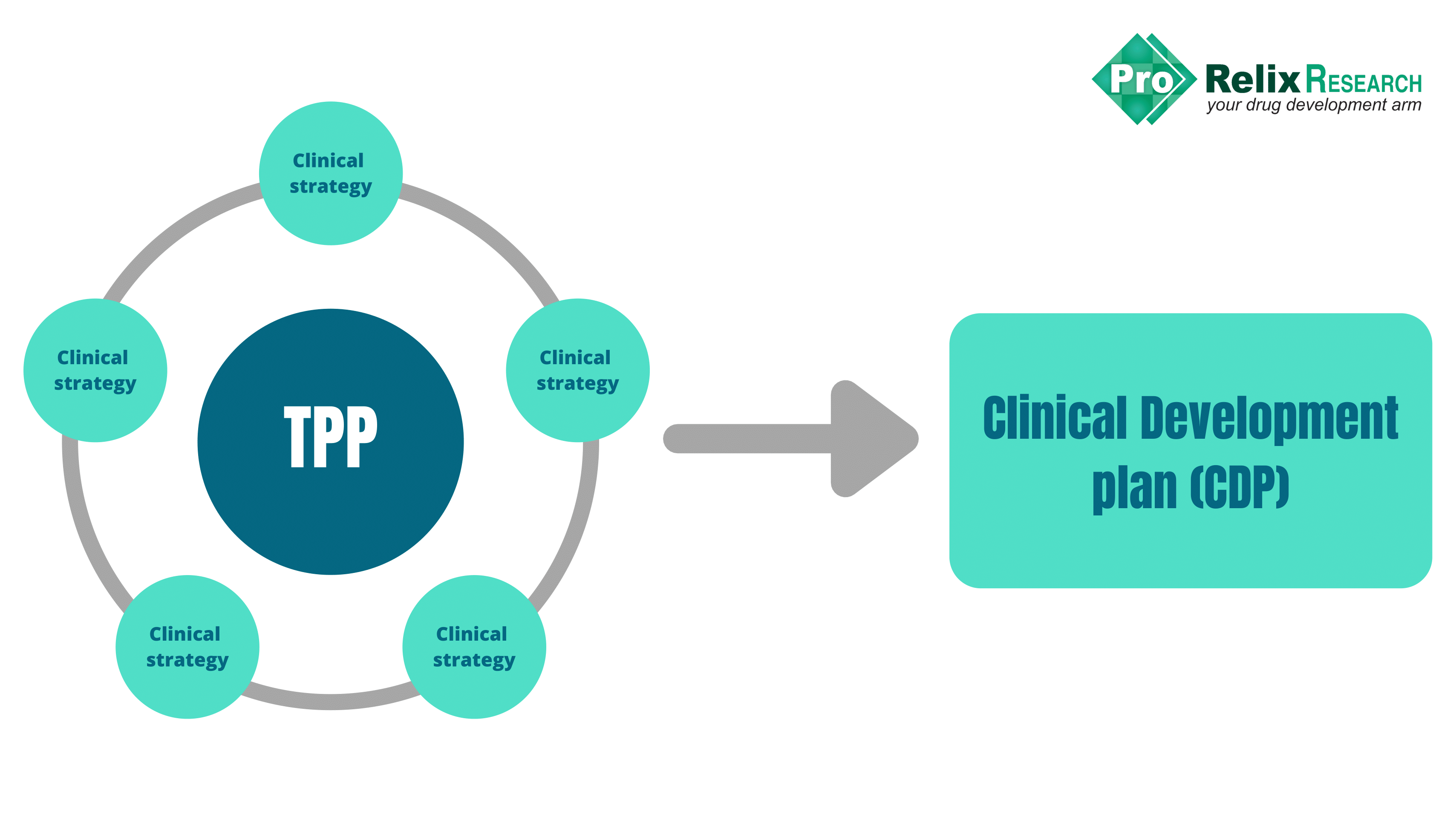
Drug development strategy for US FDA - ProRelix Research

PDF) What's the Regulatory Value of a Target Product Profile?
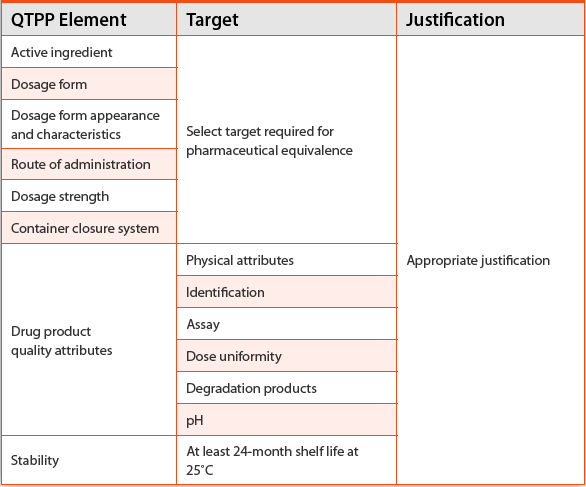
Quality by Design (QbD) Approach to Generic Transdermal or Topical Product Development American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Drug Development Strategy for US FDA

Nelson Kinnersley on LinkedIn: The Value of a Target Product Profile (TPP)










